Description
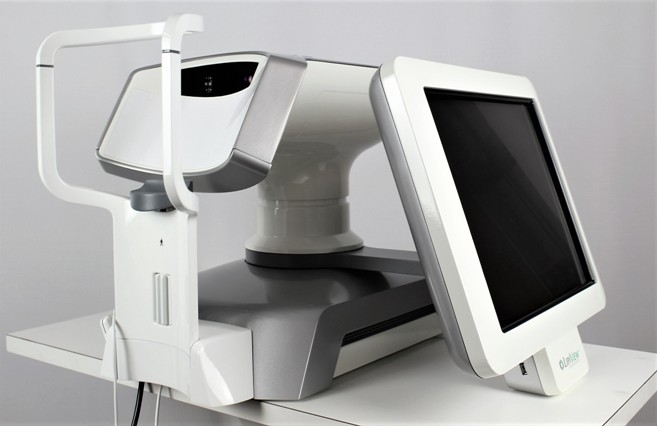
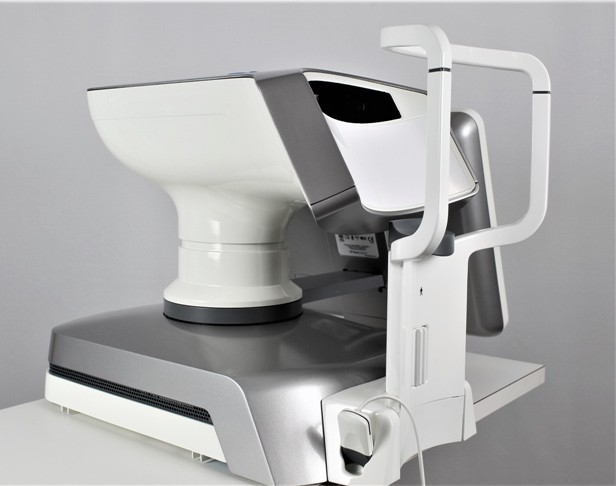
LipiView II Ocular Surface Interferometer
The LipiView II Ocular Surface Interferometer is an ophthalmic imaging device that is intended for use by a physician in adult patients to capture, archive, manipulate and store digital images of:
• Specular (interferometric) observations of the tear film. Using these images, LipiView II measures the absolute thickness of the tear film lipid layer.
• Meibomian glands under near-infrared (NIR) illumination.
• The ocular surface and eyelids under white illumination.
LipiView II Ocular Surface Interferometer Features
- Real-time visualization of the lipid layer to evaluate the dynamic response of lipids to blinking
- Patented noise canceling technology to measure sub-micron thickness of the lipid layer
- Video and analysis of blink dynamics
- High-definition imaging with Dynamic Meibomian Imaging
- Analyzes blink patterns and detects partial blinks. A graphical representation shows fluctuations in lipid layer thickness measurements between each blink
- Utilizes advanced illumination technology to capture high-definition images
- Presents lipid layer thickness measurements in an easy to understand color-coded map
CONTRAINDICATIONS
Contraindications are conditions in which the device should not be used because the risk of use clearly outweighs any benefit. No contraindications have been identified for LipiView II.
PRECAUTIONS
The following patient conditions may affect the interferometry assessment of a patient’s tear film using LipiView II:
• Use of ophthalmic drops such as artificial tear lubricants, ointments, and medications.
Advise patients not not instill oil-based ophthalmic drops (e.g., Soothe ®, Restasis®, Systane Balance®) for at least 12 hours prior to device use and not to instill ointments for at least 24 hours prior to device use. Wait at least four (4)
hours after the instillation of all other ophthalmic drops prior to device use.
• Soft or rigid contact lens wear. Advise patients to remove contact lenses at least four hours prior to device use.
• Use of oil-based facial cosmetics around the eye.
• Eye rubbing.
• Recent swimming in a chlorinated pool. Advise patients to not to swim for at least 12 hours prior to device use.
• Any ocular surface condition that affects the stability of the tear film. These conditions include disease, dystrophy, trauma, scarring, surgery, or abnormality.
Phaco & Ophthalmic Equipment Main Page